What is Erythropoietin?
Erythropoietin (EPO) is a glycoprotein hormone that controls erythopoiesis, the production of red blood cells. It also has other known biological functions; erythropoietin plays an important role in the brain's response to neuronal injury.EPO is also involved in the wound healing process. It is a cytokine for red blood cell precursors in the bone marrow._
It is produced by interstitial fibroblasts in the kidney in close association with peritubular capillary and tubular epithelial cells. It is also produced in perisinusoidal Ito cells in the liver. Erythropoietin is produced at a lesser extent by the liver. Approximately about 10% of erythropoietin is produced in the liver and the other 90% in the kidneys. The erythropoietin gene has been found on human chromosome 7 (in band 7q21). The different DNA around erythropoietin gene act to control the production of erythropoietin from the liver and of the kidney.
Kidney cells that make erythropoietin is specialized so that they are sensitive to the oxygen levels in the blood that flows through the kidneys. These cells make and release erythropoietin when oxygen levels are too low. Low oxygen levels in the kidney may indicate anemia, a number of red blood cells are reduced, or molecules of hemoglobin that carries oxygen throughout the body.
It is produced by interstitial fibroblasts in the kidney in close association with peritubular capillary and tubular epithelial cells. It is also produced in perisinusoidal Ito cells in the liver. Erythropoietin is produced at a lesser extent by the liver. Approximately about 10% of erythropoietin is produced in the liver and the other 90% in the kidneys. The erythropoietin gene has been found on human chromosome 7 (in band 7q21). The different DNA around erythropoietin gene act to control the production of erythropoietin from the liver and of the kidney.
Kidney cells that make erythropoietin is specialized so that they are sensitive to the oxygen levels in the blood that flows through the kidneys. These cells make and release erythropoietin when oxygen levels are too low. Low oxygen levels in the kidney may indicate anemia, a number of red blood cells are reduced, or molecules of hemoglobin that carries oxygen throughout the body.
Erythropoietin as the drug

_ Using recombinant DNA technology, erythropoietin has been produced
synthetically to use as a treatment for certain types of
anemia. Erythropoietin can be used to fix anemia by stimulating
red blood cell production in bone marrow.The
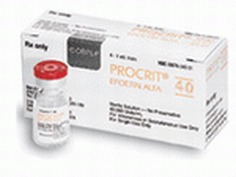
medicine is known as epoetin alfa (Epogen, Procrit). It can be given as
an injection intravenously (in the vein) or subcutaneously (under the skin).
Epoetin alfa (Epogen, Procrit)] is used in many installation-fitting clinic. The most common use is in people with anemia associated with abnormal function of the kidney. When the kidneys are not functioning properly, they produce less than normal amounts of erythropoietin, which can lead to the production of red blood cells being low, or anemia. Therefore, by replacing erythropoietin with an injection of synthetic erythropoietin, anemia associated with kidney disease may be treated. Today, Epogen or Procrit is a standard part of therapy in patients with kidney disease who require dialysis to both treat and prevent anemia.Other uses of erythropoietin may include treatment of anemia associated with AZT treatment (used to treat AIDS) and cancer-related anemia. EPO has been shown to also help certain neurological diseases, like schizophrenia. Research has also suggested that EPO improves the survival rate for children suffering from cerebral malaria; which is caused by malaria parasite's blocking blood vessels in the brain. Epo has also been used by athletes, most notoriously in the Tour De France bicycle racers, to increase their endurance. In addition, recent studies have suggested that overuse of the drug can harm patients. Sales of this drug, as a result, have declined over the last few years.
Epoetin alfa (Epogen, Procrit)] is used in many installation-fitting clinic. The most common use is in people with anemia associated with abnormal function of the kidney. When the kidneys are not functioning properly, they produce less than normal amounts of erythropoietin, which can lead to the production of red blood cells being low, or anemia. Therefore, by replacing erythropoietin with an injection of synthetic erythropoietin, anemia associated with kidney disease may be treated. Today, Epogen or Procrit is a standard part of therapy in patients with kidney disease who require dialysis to both treat and prevent anemia.Other uses of erythropoietin may include treatment of anemia associated with AZT treatment (used to treat AIDS) and cancer-related anemia. EPO has been shown to also help certain neurological diseases, like schizophrenia. Research has also suggested that EPO improves the survival rate for children suffering from cerebral malaria; which is caused by malaria parasite's blocking blood vessels in the brain. Epo has also been used by athletes, most notoriously in the Tour De France bicycle racers, to increase their endurance. In addition, recent studies have suggested that overuse of the drug can harm patients. Sales of this drug, as a result, have declined over the last few years.
Discovery of Erythropoietin

_ In the late 1970s, Dr. Goldwasser, working at the University of Chicago, isolated and purified erythropoietin aka Epo. He then shared his findings with a young biotechnology company,
which figured out how to produce larger amounts of the protein using
genetic engineering.
That company, Amgen,
became the world’s biggest biotechnology company on the basis of Epo.
Sales of erythropoietin under names like Epogen, Procrit and Aranesp amounted
to billions of dollars a year for Amgen, as well as Johnson & Johnson and Roche.
Most people undergoing kidney dialysis now receive Epo, helping to relieve them of severe anemia, which can sap them of energy. Many cancer patients also get the drug to combat anemia caused by chemotherapy.
While Epo created huge profits for drug companies, Dr. Goldwasser won neither fame nor fortune. Although he notified his university about his accomplishment, it never patented Epo, and Dr. Goldwasser did not follow up. Years after this he told a university publicist “One percent of one percent of the drug’s annual revenues would have funded my lab quite handsomely.”
As far back as 1906, two French researchers had postulated the existence of a substance that prompts the production of red blood cells, which carry oxygen to the body’s tissues. But if that substance did exist, it was in such minuscule quantities that no one could find it.
Dr. Goldwasser began to look for it in 1955 at the urging of his mentor, the noted hematologist Leon O. Jacobson. Originally he had thought it would take only several months to accomplish this, but instead it took 20 years. In 1957, Dr. Goldwasser and colleagues found out that Epo was made in the kidneys by systematically removing different organs from rats to see if they became anemic. That helped explain why patients with kidney failure became anemic. He figured that animals with anemia would produce more Epo, making finding the protein much easier to find. Dr. Goldwasser then spent years visiting a slaughterhouse outside Chicago, there he would inject sheep with a chemical that would make them anemic. Afterwards he would collect the blood and try to separate out the various components to find erythropoietin.
It ended up easier to find Epo in urine than in blood. In 1973, when his search seemed to be going nowhere, Dr. Goldwasser received a letter from Takaji Miyake of Kumamoto University in Japan. Takaji Miyake had been collecting urine from people with a disease called aplastic anemia. At the end of 1975, Dr. Miyake and Dr. Goldwasser met at Palmer House hotel in Chicago. Dr. Miyake gave Dr. Golwasser a foot-square package wrapped in brightly colored silk, inside was the dried concentrate of 2,550 liters, or about 674 gallons, of urine. From that material Dr. Goldwasser and his assistant Charles Kung and Dr. Miyake were able to purify 8 milligrams, about 3 ten-thousandths of an ounce, of Epo. Just enough to fill a small vial. They then published a paper in 1977. This difficult process of extraction was not practical for producing enough Epo to use as a drug, but the age of gene splicing was dawning. Getting some of the protein’s composition from Dr. Golwasser, Fu-Kuen Lin, a scientist at Amgen, was eventually able to clone the human gene for Epo. The gene was spliced into hamster cells, which churned out enough Epo to sell as a drug.
This drug was first tested by patients undergoing dialysis, who suffered debilitating anemia. The only treatment at that time, before Epo, was frequent blood transfusions. Dr. John W. Adamson conducted the first trial around 1985 while at the University of Washington. Patients who before had so little energy they could barely walk were now fully functional, Dr. Adamson said. Nowadays, thanks to Epo, patients with such a horrible anemic dialysis have dissapeared accoridng to Dr. Adamson, who is now at the University of California, San Diego.
Most people undergoing kidney dialysis now receive Epo, helping to relieve them of severe anemia, which can sap them of energy. Many cancer patients also get the drug to combat anemia caused by chemotherapy.
While Epo created huge profits for drug companies, Dr. Goldwasser won neither fame nor fortune. Although he notified his university about his accomplishment, it never patented Epo, and Dr. Goldwasser did not follow up. Years after this he told a university publicist “One percent of one percent of the drug’s annual revenues would have funded my lab quite handsomely.”
As far back as 1906, two French researchers had postulated the existence of a substance that prompts the production of red blood cells, which carry oxygen to the body’s tissues. But if that substance did exist, it was in such minuscule quantities that no one could find it.
Dr. Goldwasser began to look for it in 1955 at the urging of his mentor, the noted hematologist Leon O. Jacobson. Originally he had thought it would take only several months to accomplish this, but instead it took 20 years. In 1957, Dr. Goldwasser and colleagues found out that Epo was made in the kidneys by systematically removing different organs from rats to see if they became anemic. That helped explain why patients with kidney failure became anemic. He figured that animals with anemia would produce more Epo, making finding the protein much easier to find. Dr. Goldwasser then spent years visiting a slaughterhouse outside Chicago, there he would inject sheep with a chemical that would make them anemic. Afterwards he would collect the blood and try to separate out the various components to find erythropoietin.
It ended up easier to find Epo in urine than in blood. In 1973, when his search seemed to be going nowhere, Dr. Goldwasser received a letter from Takaji Miyake of Kumamoto University in Japan. Takaji Miyake had been collecting urine from people with a disease called aplastic anemia. At the end of 1975, Dr. Miyake and Dr. Goldwasser met at Palmer House hotel in Chicago. Dr. Miyake gave Dr. Golwasser a foot-square package wrapped in brightly colored silk, inside was the dried concentrate of 2,550 liters, or about 674 gallons, of urine. From that material Dr. Goldwasser and his assistant Charles Kung and Dr. Miyake were able to purify 8 milligrams, about 3 ten-thousandths of an ounce, of Epo. Just enough to fill a small vial. They then published a paper in 1977. This difficult process of extraction was not practical for producing enough Epo to use as a drug, but the age of gene splicing was dawning. Getting some of the protein’s composition from Dr. Golwasser, Fu-Kuen Lin, a scientist at Amgen, was eventually able to clone the human gene for Epo. The gene was spliced into hamster cells, which churned out enough Epo to sell as a drug.
This drug was first tested by patients undergoing dialysis, who suffered debilitating anemia. The only treatment at that time, before Epo, was frequent blood transfusions. Dr. John W. Adamson conducted the first trial around 1985 while at the University of Washington. Patients who before had so little energy they could barely walk were now fully functional, Dr. Adamson said. Nowadays, thanks to Epo, patients with such a horrible anemic dialysis have dissapeared accoridng to Dr. Adamson, who is now at the University of California, San Diego.
Chemistry Quick Facts
The chemical formula of Erythropoietin is C815H1317N233O241S5
Melting point of 53 ˚C
Molecular weight 18396 g/mol__
Melting point of 53 ˚C
Molecular weight 18396 g/mol__
Chemical Structure
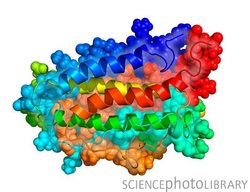
_Erythrpoietin is made up of a four-membered alpha-helical bundle with connecting loops
Although its nucleotide and amino acid sequences are known by scientists, its tertiary structure remains elusive. Antipeptide antibody and mutagenesis studies have increased the ability to be able to study structure/function and certain spatial relationships.
Although its nucleotide and amino acid sequences are known by scientists, its tertiary structure remains elusive. Antipeptide antibody and mutagenesis studies have increased the ability to be able to study structure/function and certain spatial relationships.
References
_Siren AL et al. (2001). "Erythropoietin prevents neuronal apoptosis after cerebral ischemia and metabolic stress". Proc Natl Acad Sci USA 98 (7): 4044–4049. doi:10.1073/pnas.051606598. PMC 31176. PMID 11259643.
Haroon ZA, Amin K, Jiang X, Arcasoy MO (September 2003). "A novel role for erythropoietin during fibrin-induced wound-healing response". Am. J. Pathol. 163 (3): 993–1000. PMC 1868246. PMID 12937140.
Jelkmann W (March 2007). "Erythropoietin after a century of research: younger than ever". European journal of haematology 78 (3): 183–205. doi:10.1111/j.1600-0609.2007.00818.x. PMID 17253966.
Ahmet Höke (2005). Erythropoietin and the Nervous System. Berlin: Springer. ISBN 0-387-30010-4. OCLC 64571745.
Miyake T; Kung, CK; Goldwasser, E (Aug 1997). "Purification of human erythropoietin". J. Biol. Chem. 252 (15): 5558–5564. PMID 18467.
Jelkmann W (March 2007). "Erythropoietin after a century of research: younger than ever". Eur. J. Haematol. 78 (3): 183–205. doi:10.1111/j.1600-0609.2007.00818.x. PMID 17253966.
Angell, Marcia (2005). The Truth About the Drug Companies : How They Deceive Us and What to Do About It. New York: Random House Trade Paperbacks. p. 60. ISBN 0-375-76094-6.
Eschbach JW, Egrie JC, Downing MR, Browne JK, Adamson JW (January 1987). "Correction of the anemia of end-stage renal disease with recombinant human erythropoietin. Results of a combined phase I and II clinical trial". N. Engl. J. Med. 316 (2): 73–8. doi:10.1056/NEJM198701083160203. PMID 3537801.
Lin FK, Suggs S, Lin CH, Browne JK, Smalling R, Egrie JC, Chen KK, Fox GM, Martin F, Stabinsky Z (November 1985). "Cloning and expression of the human erythropoietin gene". Proc. Natl. Acad. Sci. U.S.A. 82 (22): 7580–4. doi:10.1073/pnas.82.22.7580. PMC 391376. PMID 3865178.
Macdougall IC (July 2000). "Novel erythropoiesis stimulating protein". Semin. Nephrol. 20 (4): 375–81. PMID 10928340.
Jacobson LO, Goldwasser E, Fried W, Plzak L (March 1957). "Role of the kidney in erythropoiesis". Nature 179 (4560): 633–4. doi:10.1038/179633a0. PMID 13418752.
Fisher JW, Koury S, Ducey T, Mendel S (October 1996). "Erythropoietin production by interstitial cells of hypoxic monkey kidneys". British journal of haematology 95 (1): 27–32. doi:10.1046/j.1365-2141.1996.d01-1864.x. PMID 8857934.
Ashby DR, Gale DP, Busbridge M, et al. (March 2010). "Erythropoietin administration in humans causes a marked and prolonged reduction in circulating hepcidin". Haematologica 95 (3): 505–8. doi:10.3324/haematol.2009.013136. PMC 2833083. PMID 19833632.
Macdougall IC, Tucker B, Thompson J, Tomson CR, Baker LR, Raine AE (1996). "A randomized controlled study of iron supplementation in patients treated with erythropoietin". Kidney Int. 50 (5): 1694–9. doi:10.1038/ki.1996.487. PMID 8914038.
Smith A (2008-03-13). "FDA panel gives surprise OK to Amgen and J&J: FDA panelists support keeping Amgen, J&J drugs on market - Mar. 13, 2008". CNNMoney.com. Retrieved 2009-03-31.
"Procrit (Epoetin alfa)". Ortho Biotech Products. Retrieved 2009-04-29.
"Aranesp(darbepoetin alfa)". Amgen.com. Retrieved 2009-04-29.
Corwin HL, Gettinger A, Fabian TC, May A, Pearl RG, Heard S, An R, Bowers PJ, Burton P, Klausner MA, Corwin MJ (September 2007). "Efficacy and safety of epoetin alfa in critically ill patients". The New England Journal of Medicine 357 (10): 965–76. doi:10.1056/NEJMoa071533. PMID 17804841.
Yoo YC, Shim JK, Kim JC, Jo YY, Lee JH, Kwak YL (November 2011). "Effect of single recombinant human erythropoietin injection on transfusion requirements in preoperatively anemic patients undergoing valvular heart surgery". Anesthesiology 115 (5): 929–37. doi:10.1097/ALN.0b013e318232004b. PMID 22027622. Lay summary – Anesthesiology's blog for education & more.
Steeg JL (2007-02-28). "Catlin has made a career out of busting juicers - USATODAY.com". USA TODAY. Retrieved 2009-03-31.
Lasne F, Martin L, Crepin N, de Ceaurriz J (December 2002). "Detection of isoelectric profiles of erythropoietin in urine: differentiation of natural and administered recombinant hormones". Anal. Biochem. 311 (2): 119–26. doi:10.1016/S0003-2697(02)00407-4. PMID 12470670.
Kohler M, Ayotte C, Desharnais P, Flenker U, Lüdke S, Thevis M, Völker-Schänzer E, Schänzer W (January 2008). "Discrimination of recombinant and endogenous urinary erythropoietin by calculating relative mobility values from SDS gels". Int J Sports Med 29 (1): 1–6. doi:10.1055/s-2007-989369. PMID 18050057.
Ehrenreich H, Degner D, Meller J, et al. (January 2004). "Erythropoietin: a candidate compound for neuroprotection in schizophrenia" (PDF). Molecular psychiatry 9 (1): 42–54. doi:10.1038/sj.mp.4001442. PMID 14581931.
Casals-Pascual C, Idro R, Picot S, Roberts DJ, Newton CR (2009). "Can erythropoietin be used to prevent brain damage in cerebral malaria?". Trends Parasitol 25 (1): 30–6. doi:10.1016/j.pt.2008.10.002. PMID 19008152.
Core A, Hempel C, Kurtzhals JA, Penkowa M (2011). "Plasmodium berghei ANKA: erythropoietin activates neural stem cells in an experimental cerebral malaria model.". Exp Parasitol 127 (2): 500–5. doi:10.1016/j.exppara.2010.09.010. PMID 21044627.
"Kidney drug could save children from malaria brain damage". The Guardian.
Swartjes M, Morariu A, Niesters M, Brines M, Cerami A, Aarts L, Dahan A (November 2011). "ARA290, a Peptide Derived from the Tertiary Structure of Erythropoietin, Produces Long-term Relief of Neuropathic Pain: An Experimental Study in Rats and β-Common Receptor Knockout Mice". Anesthesiology 115 (5): 1084–92. doi:10.1097/ALN.0b013e31822fcefd. PMID 21873879. Lay summary – Anesthesiology's blog for education & more.
Drüeke TB, Locatelli F, Clyne N, Eckardt KU, Macdougall IC, Tsakiris D, Burger HU, Scherhag A (2006). "Normalization of hemoglobin level in patients with chronic kidney disease and anemia". N. Engl. J. Med. 355 (20): 2071–84. doi:10.1056/NEJMoa062276. PMID 17108342.
Ohlsson A, Aher SM (2006). "Early erythropoietin for preventing red blood cell transfusion in preterm and/or low birth weight infants". Cochrane Database Syst Rev 3: CD004863. doi:10.1002/14651858.CD004863.pub2. PMID 16856062.
Aher SM, Ohlsson A (2006). "Early versus late erythropoietin for preventing red blood cell transfusion in preterm and/or low birth weight infants". Cochrane Database Syst Rev 3: CD004865. doi:10.1002/14651858.CD004865.pub2. PMID 16856063.
"FDA Public Health Advisory: Erythropoiesis-Stimulating Agents (ESAs): Epoetin alfa (marketed as Procrit, Epogen), Darbepoetin alfa (marketed as Aranesp)". Archived from the original on 2007-05-28. Retrieved 2007-06-05.
"Information for Healthcare Professionals: Erythropoiesis Stimulating Agents (ESA)". Archived from the original on 2007-05-15. Retrieved 2007-06-05.
"Erythropoiesis Stimulating Agents: Aranesp (darbepoetin alfa), Epogen (epoetin alfa), and Procrit (epoetin alfa)". MedWatch - 2007 Safety Information Alerts. U.S. Food and Drug Administration. 2008-01-03. Retrieved 2009-04-09.
"Procrit (Epoetin alfa) for injection". U.S. Food and Drug Administration. 2007-08-11. Retrieved 2009-04-09.
"Aranesp (darbepoetin alfa) for Injection". U.S. Food and Drug Administration. 2007-11-08. Retrieved 2009-04-09.
"Information on Erythropoiesis Stimulating Agents (ESA) (marketed as Procrit, Epogen, and Aranesp)". U.S. Food and Drug Administration. 2009-01-26. Retrieved 2009-04-09.
Middleton, S A; Barbone F P, Johnson D L, Thurmond R L, You Y, McMahon F J, Jin R, Livnah O, Tullai J, Farrell F X, Goldsmith M A, Wilson I A, Jolliffe L K (May. 1999). "Shared and unique determinants of the erythropoietin (EPO) receptor are important for binding EPO and EPO mimetic peptide". J. Biol. Chem. (UNITED STATES) 274 (20): 14163–9. doi:10.1074/jbc.274.20.14163. ISSN 0021-9258. PMID 10318834.
Livnah, O; Johnson D L, Stura E A, Farrell F X, Barbone F P, You Y, Liu K D, Goldsmith M A, He W, Krause C D, Pestka S, Jolliffe L K, Wilson I A (Nov. 1998). "An antagonist peptide-EPO receptor complex suggests that receptor dimerization is not sufficient for activation". Nat. Struct. Biol. (UNITED STATES) 5 (11): 993–1004. doi:10.1038/2965. ISSN 1072-8368. PMID 9808045.
Drug Interactions of Erythropoietin Alfa at Drugs.com
Gilman A, Goodman LS, Hardman JG, Limbird LE (2001). Goodman & Gilman's the pharmacological basis of therapeutics. New York: McGraw-Hill. ISBN 0-07-135469-7.
Boyle, J. J. (2005). Macrophage activation in atherosclerosis: pathogenesis and pharmacology of plaque rupture. Curr Vasc Pharmacol, 3(1), 63-68. PMID=15638783
Cannon JG (2000). "Inflammatory Cytokines in Nonpathological States". News Physiol Sci. 15: 298–303. PMID 11390930.
Saito, Shigeru (October–November 2001). "Cytokine cross-talk between mother and the embryo/placenta". Journal of Reproductive Immunology 52 (1–2): 15–33. doi:10.1016/S0165-0378(01)00112-7. PMID 11600175. Retrieved 2010-03-29.
Chen M.D., Hsin-Fu; Jin-Yuh Shew, Hong-Nerng Ho, Wei-Li Hsu, Yu-Shih Yang (October 1999). "Expression of leukemia inhibitory factor and its receptor in preimplantation embryos". Journal of Reproductive Biology 72 (4): 713–719. doi:10.1016/S0015-0282(99)00306-4. Retrieved 2010-03-29.
Elias A. Said et al. 2009, PD-1 Induced IL10 Production by Monocytes Impairs T-cell Activation in a Reversible Fashion. Nature Medicine. 2010; 452-9.
Vlahopoulos, S; Boldogh, I; Casola, A; Brasier, AR (1999). "Nuclear factor-kappaB-dependent induction of interleukin-8 gene expression by tumor necrosis factor alpha: evidence for an antioxidant sensitive activating pathway distinct from nuclear translocation". Blood 94 (6): 1878–89. PMID 10477716.
David, F; Farley, J; Huang, H; Lavoie, JP; Laverty, S (2007). "Cytokine and chemokine gene expression of IL-1beta stimulated equine articular chondrocytes". Veterinary surgery : VS 36 (3): 221–7. doi:10.1111/j.1532-950X.2007.00253.x. PMID 17461946.
Carpenter, LR; Moy, JN; Roebuck, KA (2002). "Respiratory syncytial virus and TNF alpha induction of chemokine gene expression involves differential activation of Rel A and NF-kappa B1". BMC infectious diseases 2: 5. PMC 102322. PMID 11922866.
Tian, B; Nowak, DE; Brasier, AR (2005). "A TNF-induced gene expression program under oscillatory NF-kappaB control". BMC genomics 6: 137. doi:10.1186/1471-2164-6-137. PMC 1262712. PMID 16191192.
Dowlati Y, Herrmann N, Swardfager W, Liu H, Sham L, Reim EK, Lanctôt KL (2010). "A meta-analysis of cytokines in major depression". Biol Psychiatry 67 (5): 446–457. doi:10.1016/j.biopsych.2009.09.033. PMID 20015486.
Swardfager W, Lanctôt K, Rothenburg L, Wong A, Cappell J, Herrmann N (2010). "A meta-analysis of cytokines in Alzheimer's disease". Biol Psychiatry 68 (10): 930–941. doi:10.1016/j.biopsych.2010.06.012. PMID 20692646.
Locksley RM, Killeen N, Lenardo MJ (2001). "The TNF and TNF receptor superfamilies: integrating mammalian biology". Cell 104 (4): 487–501. doi:10.1016/S0092-8674(01)00237-9. PMID 11239407.
Kokkonen, H. Arthritis & Rheumatism, Feb. 2, 2010; vol 62: pp 383-391
Nikolaeva LG, Maystat TV, Masyuk LA, Pylypchuk VS, Volyanskii YL, Kutsyna GA (2009). "Changes in CD4+ T-cells and HIV RNA resulting from combination of anti-TB therapy with Dzherelo in TB/HIV dually infected patients". Drug Des Devel Ther 2: 87–93. PMC 2761183. PMID 19920896.
Napolitano LA, Grant RM, Deeks SG, et al. (January 2001). "Increased production of IL-7 accompanies HIV-1-mediated T-cell depletion: implications for T-cell homeostasis". Nat. Med. 7 (1): 73–9. doi:10.1038/83381. PMID 11135619.
Weissmanshomer, P.; Fry, M. (1975). "Chick embryo fibroblasts senescence in vitro: Pattern of cell division and life span as a function of cell density". Mechanisms of Ageing and Development 4 (2): 159. doi:10.1016/0047-6374(75)90017-2. PMID 1152547.
EPO and a needle. Digital image. World of Health-knowledge. Web. 3 Dec. 2011. <http://4.bp.blogspot.com/-uACmfbsA56g/TnlXpN-mBfI/AAAAAAAABEw/vHh-ryKnW2A/s400/Erythropoietin+%2528EPO%2529++and+EPO+Test.jpg>.
"Erythropoietin (EPO) and EPO Test - World of Health-World of Health." World of Health-knowledge. Web. 03 Dec. 2011. <http://topworldofhealth.blogspot.com/2011/09/erythropoietin-epo-and-epo-test.html>.
"Fibroblast - Glossary Entry - Genetics Home Reference." Genetics Home Reference - Your Guide to Understanding Genetic Conditions. Web. 03 Dec. 2011. <http://ghr.nlm.nih.gov/glossary=fibroblast>.
Pink cells. Digital image. NASA. Web. 3 Dec. 2011. <http://www.nasa.gov/images/content/548053main_CBOSS-02-Erythropoietin1.jpg>.
Pollack, Andrew. "Eugene Goldwasser, Developer of Anemia Drug, Dies at 88 - NYTimes.com." The New York Times - Breaking News, World News & Multimedia. 20 Dec. 2010. Web. 03 Dec. 2011. <http://www.nytimes.com/2010/12/21/health/21goldwasser.html>.
Prod. Shaziajafrey8. How To Give Erythropoietin Injection? Youtube, 5 Nov. 2011. Web. 3 Dec. 2011. <http://www.youtube.com/watch?v=5KpR7i27Tlw>.
Red blood cells. Digital image. Marine Genetic. Web. 3 Dec. 2011. <http://www.marine-genetic-resources.com/uploadfile/201002/24/03811219.jpg>.
Sirén, Anna-Leena, Maddalena Fratelli, Michael Brines, Christoph Goemans, Simona Casagrande, Piotr Lewczuk, Sonja Keenan, Christoph Gleiter, Claudio Pasquali, Annalisa Capobianco, Tiziana Mennini, Rolf Heumann, Anthony Cerami, Hannelore Ehrenreich, and Pietro Ghezzi. "Erythropoietin Prevents Neuronal Apoptosis after Cerebral Ischemia and Metabolic Stress." PNAS. 21 Dec. 200. Web. 3 Dec. 2011. <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC31176/?tool=pmcentrez>.
Haroon ZA, Amin K, Jiang X, Arcasoy MO (September 2003). "A novel role for erythropoietin during fibrin-induced wound-healing response". Am. J. Pathol. 163 (3): 993–1000. PMC 1868246. PMID 12937140.
Jelkmann W (March 2007). "Erythropoietin after a century of research: younger than ever". European journal of haematology 78 (3): 183–205. doi:10.1111/j.1600-0609.2007.00818.x. PMID 17253966.
Ahmet Höke (2005). Erythropoietin and the Nervous System. Berlin: Springer. ISBN 0-387-30010-4. OCLC 64571745.
Miyake T; Kung, CK; Goldwasser, E (Aug 1997). "Purification of human erythropoietin". J. Biol. Chem. 252 (15): 5558–5564. PMID 18467.
Jelkmann W (March 2007). "Erythropoietin after a century of research: younger than ever". Eur. J. Haematol. 78 (3): 183–205. doi:10.1111/j.1600-0609.2007.00818.x. PMID 17253966.
Angell, Marcia (2005). The Truth About the Drug Companies : How They Deceive Us and What to Do About It. New York: Random House Trade Paperbacks. p. 60. ISBN 0-375-76094-6.
Eschbach JW, Egrie JC, Downing MR, Browne JK, Adamson JW (January 1987). "Correction of the anemia of end-stage renal disease with recombinant human erythropoietin. Results of a combined phase I and II clinical trial". N. Engl. J. Med. 316 (2): 73–8. doi:10.1056/NEJM198701083160203. PMID 3537801.
Lin FK, Suggs S, Lin CH, Browne JK, Smalling R, Egrie JC, Chen KK, Fox GM, Martin F, Stabinsky Z (November 1985). "Cloning and expression of the human erythropoietin gene". Proc. Natl. Acad. Sci. U.S.A. 82 (22): 7580–4. doi:10.1073/pnas.82.22.7580. PMC 391376. PMID 3865178.
Macdougall IC (July 2000). "Novel erythropoiesis stimulating protein". Semin. Nephrol. 20 (4): 375–81. PMID 10928340.
Jacobson LO, Goldwasser E, Fried W, Plzak L (March 1957). "Role of the kidney in erythropoiesis". Nature 179 (4560): 633–4. doi:10.1038/179633a0. PMID 13418752.
Fisher JW, Koury S, Ducey T, Mendel S (October 1996). "Erythropoietin production by interstitial cells of hypoxic monkey kidneys". British journal of haematology 95 (1): 27–32. doi:10.1046/j.1365-2141.1996.d01-1864.x. PMID 8857934.
Ashby DR, Gale DP, Busbridge M, et al. (March 2010). "Erythropoietin administration in humans causes a marked and prolonged reduction in circulating hepcidin". Haematologica 95 (3): 505–8. doi:10.3324/haematol.2009.013136. PMC 2833083. PMID 19833632.
Macdougall IC, Tucker B, Thompson J, Tomson CR, Baker LR, Raine AE (1996). "A randomized controlled study of iron supplementation in patients treated with erythropoietin". Kidney Int. 50 (5): 1694–9. doi:10.1038/ki.1996.487. PMID 8914038.
Smith A (2008-03-13). "FDA panel gives surprise OK to Amgen and J&J: FDA panelists support keeping Amgen, J&J drugs on market - Mar. 13, 2008". CNNMoney.com. Retrieved 2009-03-31.
"Procrit (Epoetin alfa)". Ortho Biotech Products. Retrieved 2009-04-29.
"Aranesp(darbepoetin alfa)". Amgen.com. Retrieved 2009-04-29.
Corwin HL, Gettinger A, Fabian TC, May A, Pearl RG, Heard S, An R, Bowers PJ, Burton P, Klausner MA, Corwin MJ (September 2007). "Efficacy and safety of epoetin alfa in critically ill patients". The New England Journal of Medicine 357 (10): 965–76. doi:10.1056/NEJMoa071533. PMID 17804841.
Yoo YC, Shim JK, Kim JC, Jo YY, Lee JH, Kwak YL (November 2011). "Effect of single recombinant human erythropoietin injection on transfusion requirements in preoperatively anemic patients undergoing valvular heart surgery". Anesthesiology 115 (5): 929–37. doi:10.1097/ALN.0b013e318232004b. PMID 22027622. Lay summary – Anesthesiology's blog for education & more.
Steeg JL (2007-02-28). "Catlin has made a career out of busting juicers - USATODAY.com". USA TODAY. Retrieved 2009-03-31.
Lasne F, Martin L, Crepin N, de Ceaurriz J (December 2002). "Detection of isoelectric profiles of erythropoietin in urine: differentiation of natural and administered recombinant hormones". Anal. Biochem. 311 (2): 119–26. doi:10.1016/S0003-2697(02)00407-4. PMID 12470670.
Kohler M, Ayotte C, Desharnais P, Flenker U, Lüdke S, Thevis M, Völker-Schänzer E, Schänzer W (January 2008). "Discrimination of recombinant and endogenous urinary erythropoietin by calculating relative mobility values from SDS gels". Int J Sports Med 29 (1): 1–6. doi:10.1055/s-2007-989369. PMID 18050057.
Ehrenreich H, Degner D, Meller J, et al. (January 2004). "Erythropoietin: a candidate compound for neuroprotection in schizophrenia" (PDF). Molecular psychiatry 9 (1): 42–54. doi:10.1038/sj.mp.4001442. PMID 14581931.
Casals-Pascual C, Idro R, Picot S, Roberts DJ, Newton CR (2009). "Can erythropoietin be used to prevent brain damage in cerebral malaria?". Trends Parasitol 25 (1): 30–6. doi:10.1016/j.pt.2008.10.002. PMID 19008152.
Core A, Hempel C, Kurtzhals JA, Penkowa M (2011). "Plasmodium berghei ANKA: erythropoietin activates neural stem cells in an experimental cerebral malaria model.". Exp Parasitol 127 (2): 500–5. doi:10.1016/j.exppara.2010.09.010. PMID 21044627.
"Kidney drug could save children from malaria brain damage". The Guardian.
Swartjes M, Morariu A, Niesters M, Brines M, Cerami A, Aarts L, Dahan A (November 2011). "ARA290, a Peptide Derived from the Tertiary Structure of Erythropoietin, Produces Long-term Relief of Neuropathic Pain: An Experimental Study in Rats and β-Common Receptor Knockout Mice". Anesthesiology 115 (5): 1084–92. doi:10.1097/ALN.0b013e31822fcefd. PMID 21873879. Lay summary – Anesthesiology's blog for education & more.
Drüeke TB, Locatelli F, Clyne N, Eckardt KU, Macdougall IC, Tsakiris D, Burger HU, Scherhag A (2006). "Normalization of hemoglobin level in patients with chronic kidney disease and anemia". N. Engl. J. Med. 355 (20): 2071–84. doi:10.1056/NEJMoa062276. PMID 17108342.
Ohlsson A, Aher SM (2006). "Early erythropoietin for preventing red blood cell transfusion in preterm and/or low birth weight infants". Cochrane Database Syst Rev 3: CD004863. doi:10.1002/14651858.CD004863.pub2. PMID 16856062.
Aher SM, Ohlsson A (2006). "Early versus late erythropoietin for preventing red blood cell transfusion in preterm and/or low birth weight infants". Cochrane Database Syst Rev 3: CD004865. doi:10.1002/14651858.CD004865.pub2. PMID 16856063.
"FDA Public Health Advisory: Erythropoiesis-Stimulating Agents (ESAs): Epoetin alfa (marketed as Procrit, Epogen), Darbepoetin alfa (marketed as Aranesp)". Archived from the original on 2007-05-28. Retrieved 2007-06-05.
"Information for Healthcare Professionals: Erythropoiesis Stimulating Agents (ESA)". Archived from the original on 2007-05-15. Retrieved 2007-06-05.
"Erythropoiesis Stimulating Agents: Aranesp (darbepoetin alfa), Epogen (epoetin alfa), and Procrit (epoetin alfa)". MedWatch - 2007 Safety Information Alerts. U.S. Food and Drug Administration. 2008-01-03. Retrieved 2009-04-09.
"Procrit (Epoetin alfa) for injection". U.S. Food and Drug Administration. 2007-08-11. Retrieved 2009-04-09.
"Aranesp (darbepoetin alfa) for Injection". U.S. Food and Drug Administration. 2007-11-08. Retrieved 2009-04-09.
"Information on Erythropoiesis Stimulating Agents (ESA) (marketed as Procrit, Epogen, and Aranesp)". U.S. Food and Drug Administration. 2009-01-26. Retrieved 2009-04-09.
Middleton, S A; Barbone F P, Johnson D L, Thurmond R L, You Y, McMahon F J, Jin R, Livnah O, Tullai J, Farrell F X, Goldsmith M A, Wilson I A, Jolliffe L K (May. 1999). "Shared and unique determinants of the erythropoietin (EPO) receptor are important for binding EPO and EPO mimetic peptide". J. Biol. Chem. (UNITED STATES) 274 (20): 14163–9. doi:10.1074/jbc.274.20.14163. ISSN 0021-9258. PMID 10318834.
Livnah, O; Johnson D L, Stura E A, Farrell F X, Barbone F P, You Y, Liu K D, Goldsmith M A, He W, Krause C D, Pestka S, Jolliffe L K, Wilson I A (Nov. 1998). "An antagonist peptide-EPO receptor complex suggests that receptor dimerization is not sufficient for activation". Nat. Struct. Biol. (UNITED STATES) 5 (11): 993–1004. doi:10.1038/2965. ISSN 1072-8368. PMID 9808045.
Drug Interactions of Erythropoietin Alfa at Drugs.com
Gilman A, Goodman LS, Hardman JG, Limbird LE (2001). Goodman & Gilman's the pharmacological basis of therapeutics. New York: McGraw-Hill. ISBN 0-07-135469-7.
Boyle, J. J. (2005). Macrophage activation in atherosclerosis: pathogenesis and pharmacology of plaque rupture. Curr Vasc Pharmacol, 3(1), 63-68. PMID=15638783
Cannon JG (2000). "Inflammatory Cytokines in Nonpathological States". News Physiol Sci. 15: 298–303. PMID 11390930.
Saito, Shigeru (October–November 2001). "Cytokine cross-talk between mother and the embryo/placenta". Journal of Reproductive Immunology 52 (1–2): 15–33. doi:10.1016/S0165-0378(01)00112-7. PMID 11600175. Retrieved 2010-03-29.
Chen M.D., Hsin-Fu; Jin-Yuh Shew, Hong-Nerng Ho, Wei-Li Hsu, Yu-Shih Yang (October 1999). "Expression of leukemia inhibitory factor and its receptor in preimplantation embryos". Journal of Reproductive Biology 72 (4): 713–719. doi:10.1016/S0015-0282(99)00306-4. Retrieved 2010-03-29.
Elias A. Said et al. 2009, PD-1 Induced IL10 Production by Monocytes Impairs T-cell Activation in a Reversible Fashion. Nature Medicine. 2010; 452-9.
Vlahopoulos, S; Boldogh, I; Casola, A; Brasier, AR (1999). "Nuclear factor-kappaB-dependent induction of interleukin-8 gene expression by tumor necrosis factor alpha: evidence for an antioxidant sensitive activating pathway distinct from nuclear translocation". Blood 94 (6): 1878–89. PMID 10477716.
David, F; Farley, J; Huang, H; Lavoie, JP; Laverty, S (2007). "Cytokine and chemokine gene expression of IL-1beta stimulated equine articular chondrocytes". Veterinary surgery : VS 36 (3): 221–7. doi:10.1111/j.1532-950X.2007.00253.x. PMID 17461946.
Carpenter, LR; Moy, JN; Roebuck, KA (2002). "Respiratory syncytial virus and TNF alpha induction of chemokine gene expression involves differential activation of Rel A and NF-kappa B1". BMC infectious diseases 2: 5. PMC 102322. PMID 11922866.
Tian, B; Nowak, DE; Brasier, AR (2005). "A TNF-induced gene expression program under oscillatory NF-kappaB control". BMC genomics 6: 137. doi:10.1186/1471-2164-6-137. PMC 1262712. PMID 16191192.
Dowlati Y, Herrmann N, Swardfager W, Liu H, Sham L, Reim EK, Lanctôt KL (2010). "A meta-analysis of cytokines in major depression". Biol Psychiatry 67 (5): 446–457. doi:10.1016/j.biopsych.2009.09.033. PMID 20015486.
Swardfager W, Lanctôt K, Rothenburg L, Wong A, Cappell J, Herrmann N (2010). "A meta-analysis of cytokines in Alzheimer's disease". Biol Psychiatry 68 (10): 930–941. doi:10.1016/j.biopsych.2010.06.012. PMID 20692646.
Locksley RM, Killeen N, Lenardo MJ (2001). "The TNF and TNF receptor superfamilies: integrating mammalian biology". Cell 104 (4): 487–501. doi:10.1016/S0092-8674(01)00237-9. PMID 11239407.
Kokkonen, H. Arthritis & Rheumatism, Feb. 2, 2010; vol 62: pp 383-391
Nikolaeva LG, Maystat TV, Masyuk LA, Pylypchuk VS, Volyanskii YL, Kutsyna GA (2009). "Changes in CD4+ T-cells and HIV RNA resulting from combination of anti-TB therapy with Dzherelo in TB/HIV dually infected patients". Drug Des Devel Ther 2: 87–93. PMC 2761183. PMID 19920896.
Napolitano LA, Grant RM, Deeks SG, et al. (January 2001). "Increased production of IL-7 accompanies HIV-1-mediated T-cell depletion: implications for T-cell homeostasis". Nat. Med. 7 (1): 73–9. doi:10.1038/83381. PMID 11135619.
Weissmanshomer, P.; Fry, M. (1975). "Chick embryo fibroblasts senescence in vitro: Pattern of cell division and life span as a function of cell density". Mechanisms of Ageing and Development 4 (2): 159. doi:10.1016/0047-6374(75)90017-2. PMID 1152547.
EPO and a needle. Digital image. World of Health-knowledge. Web. 3 Dec. 2011. <http://4.bp.blogspot.com/-uACmfbsA56g/TnlXpN-mBfI/AAAAAAAABEw/vHh-ryKnW2A/s400/Erythropoietin+%2528EPO%2529++and+EPO+Test.jpg>.
"Erythropoietin (EPO) and EPO Test - World of Health-World of Health." World of Health-knowledge. Web. 03 Dec. 2011. <http://topworldofhealth.blogspot.com/2011/09/erythropoietin-epo-and-epo-test.html>.
"Fibroblast - Glossary Entry - Genetics Home Reference." Genetics Home Reference - Your Guide to Understanding Genetic Conditions. Web. 03 Dec. 2011. <http://ghr.nlm.nih.gov/glossary=fibroblast>.
Pink cells. Digital image. NASA. Web. 3 Dec. 2011. <http://www.nasa.gov/images/content/548053main_CBOSS-02-Erythropoietin1.jpg>.
Pollack, Andrew. "Eugene Goldwasser, Developer of Anemia Drug, Dies at 88 - NYTimes.com." The New York Times - Breaking News, World News & Multimedia. 20 Dec. 2010. Web. 03 Dec. 2011. <http://www.nytimes.com/2010/12/21/health/21goldwasser.html>.
Prod. Shaziajafrey8. How To Give Erythropoietin Injection? Youtube, 5 Nov. 2011. Web. 3 Dec. 2011. <http://www.youtube.com/watch?v=5KpR7i27Tlw>.
Red blood cells. Digital image. Marine Genetic. Web. 3 Dec. 2011. <http://www.marine-genetic-resources.com/uploadfile/201002/24/03811219.jpg>.
Sirén, Anna-Leena, Maddalena Fratelli, Michael Brines, Christoph Goemans, Simona Casagrande, Piotr Lewczuk, Sonja Keenan, Christoph Gleiter, Claudio Pasquali, Annalisa Capobianco, Tiziana Mennini, Rolf Heumann, Anthony Cerami, Hannelore Ehrenreich, and Pietro Ghezzi. "Erythropoietin Prevents Neuronal Apoptosis after Cerebral Ischemia and Metabolic Stress." PNAS. 21 Dec. 200. Web. 3 Dec. 2011. <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC31176/?tool=pmcentrez>.
